Brain in the dish: neurodevelopmental effect of radiofrequency electromagnetic fields (RF-EMF) (5G)
One research topic of the Division of Veterinary Pharmacology & Toxicology at the Vetsuisse Faculty is to explore and understand possible risks of non-ionizing radiation, namely radiofrequency electromagnetic fields (RF-EMF) exposure in the brain. We investigate effects of RF-EMF during different stages of brain development or its effect in the or development of neurodegenerative diseases (e.g., Parkinson’s disease).
Human induced pluripotent stem cell (iPSc)-derived neurons and iPSc-derived brain organoids are used as models. These types of culture require the use of an incubator to minimize humidity, temperature and pH variations during all iPSc expansion periods, induction of neuronal differentiation, and the first week of development of midbrain organoids (Figure 1).
The PHCbi CO2 incubator with four inner doors purchased with the funds provided from the Berne University Research Foundation allowed stable conditions during the culturing even though the incubator was opened various times per day due to the need to medium change etc (Figure 2).
Two projects were successfully completed:
1. Generation and characterization of iPSC-derived dopaminergic neurons to study effects of 5G radiofrequency electromagnetic fields on neuronal development
2. The role of RF-EMF (5G) on neuronal development and neuronal health using brain organoids
The obtained data showed that RF-EMF did not significantly change the investigated markers essential for neural development as well as the maturity and the dopaminergic phenotype. A non-significant trend toward changes in the ERK activation was found, indicating an effect on differentiation if it is not transient.
Preliminary results from 5G RF-EMF exposure with cerebral and midbrain organoid indicated no alteration of neuronal maturity and the dopaminergic phenotype, anda significant decrease in synaptophysin protein levels in cerebral organoids after RF-EMF exposure at day 30, indicating a reduced synaptic activity (Figures 3 and 4). Furthermore, a decrease in neuronal progenitor cells expressed in ventricle-like zones of cerebral organoids, whereas an increase of these cells was found in midbrain organoids.
Further research will include investigations at different times of midbrain development, different specific absorption rates (SAR), and exposure times during neuronal development and disease models of neurodegeneration.
Angélique Ducray, PhD
Senior Scientist
Vetsuisse Faculty
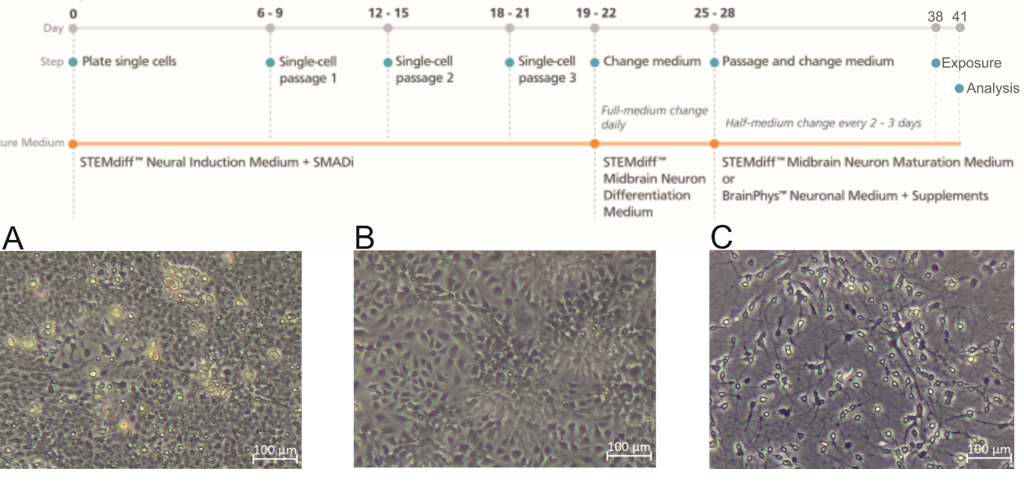
Figure 1: Timeline of monolayer production. Representative images of each of the three phases are shown. (A) Cell culture on day in vitro (DIV) 6 Passage (P) 2 during induction into neural progenitor cells (NPC). (B) Cell culture on DIV6 P3 during differentiation into midbrain neuronal precursors. (C) Cell culture on DIV9 P4 during maturation into midbrain neurons from midbrain neuronal precursors. Scale bars:100 μm.
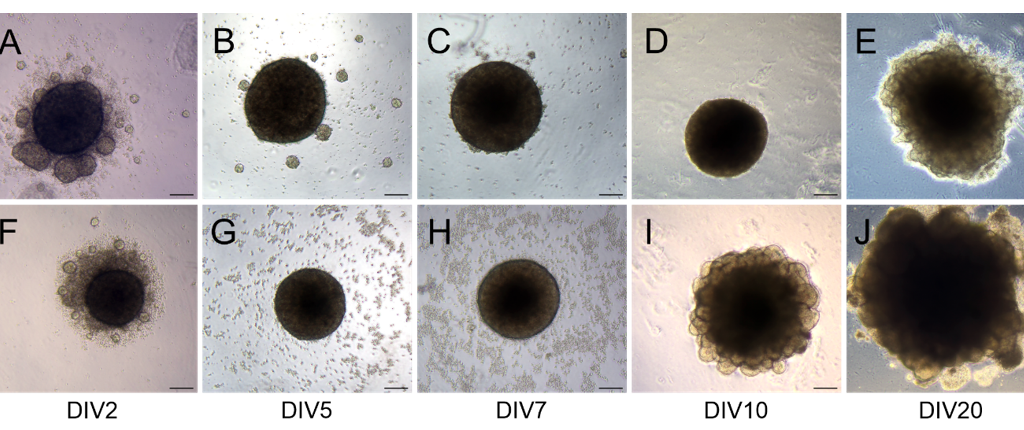
Figure 2: Homogenous development of both types of organoids showed with representative bright-field images acquired at time points DIV2, 5, 7, 10, and 20 for midbrain (A-E) and cerebral (F-J) organoids. Magnification 4x, scale bar = 200 μm.
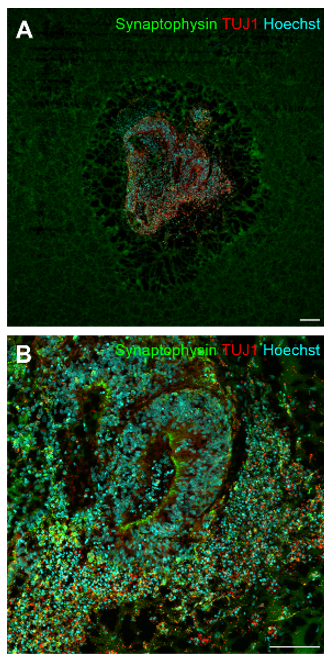
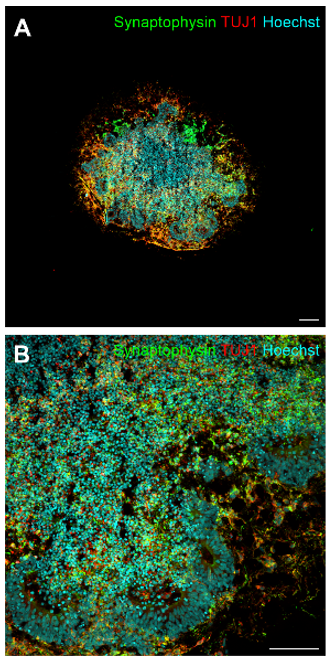
Figure 3: Representative immunofluorescence images of cerebral organoids after 30 days of development (A-B). Organoids were stained with synaptophysin (green) and TUJ1 (red). Cell nuclei were visualized with Hoechst 33342 (blue). Magnification 4x with scale bar = 200 μm (A), magnification 20x with scale bar = 100 μm (B), magnification 60x with scale bar = 20 μm
Figure 4: Representative immunofluorescence images of midbrain organoids after 30 days of development (A-B). Organoids were stained with synaptophysin (green) and TUJ1 (red). Cell nuclei were visualized with Hoechst 33342 (blue). Magnification 4x with scale bar = 200 μm (A), magnification 20x with scale bar = 100 μm (B), magnification 60x with scale bar = 20 μm